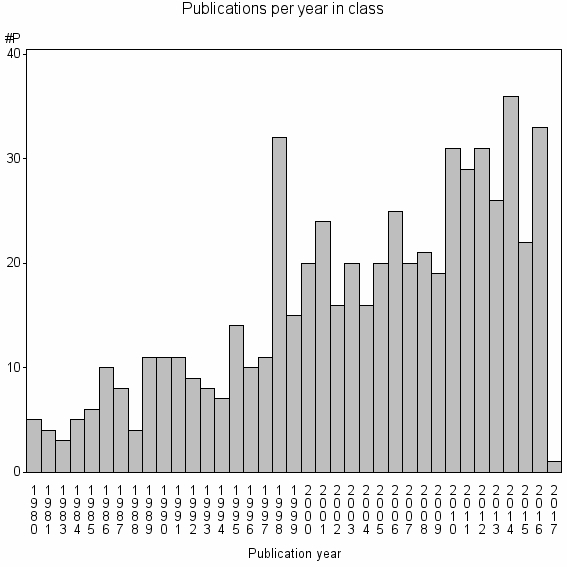
Class information for: |
Basic class information |
| Class id | #P | Avg. number of references |
Database coverage of references |
|---|---|---|---|
| 17540 | 594 | 17.7 | 48% |
Hierarchy of classes |
The table includes all classes above and classes immediately below the current class. |
Terms with highest relevance score |
| rank | Term | termType | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|---|
| 1 | RISK BASED MONITORING | authKW | 781895 | 3% | 89% | 17 |
| 2 | DOUBLE DATA ENTRY | authKW | 365546 | 1% | 89% | 8 |
| 3 | PROTOCOL COMPLEXITY | authKW | 308431 | 1% | 100% | 6 |
| 4 | CONTROLLED CLINICAL TRIALS | journal | 267016 | 12% | 7% | 71 |
| 5 | ELECTRONIC DATA CAPTURE | authKW | 261342 | 3% | 27% | 19 |
| 6 | SOURCE DATA VERIFICATION | authKW | 231320 | 1% | 75% | 6 |
| 7 | REMOTE DATA ENTRY | authKW | 228981 | 1% | 64% | 7 |
| 8 | LARGE SIMPLE TRIAL | authKW | 168229 | 1% | 55% | 6 |
| 9 | CLINICAL DATA MANAGEMENT SYSTEM | authKW | 154215 | 1% | 100% | 3 |
| 10 | TELEFORM | authKW | 154215 | 1% | 100% | 3 |
Web of Science journal categories |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | Medical Informatics | 8290 | 15% | 0% | 87 |
| 2 | Health Care Sciences & Services | 3623 | 17% | 0% | 101 |
| 3 | Pharmacology & Pharmacy | 1880 | 34% | 0% | 201 |
| 4 | Medicine, Research & Experimental | 1221 | 18% | 0% | 105 |
| 5 | Public, Environmental & Occupational Health | 363 | 11% | 0% | 67 |
| 6 | Nursing | 250 | 4% | 0% | 24 |
| 7 | Mathematical & Computational Biology | 226 | 4% | 0% | 23 |
| 8 | Computer Science, Interdisciplinary Applications | 161 | 5% | 0% | 30 |
| 9 | Statistics & Probability | 136 | 4% | 0% | 25 |
| 10 | Computer Science, Information Systems | 105 | 4% | 0% | 24 |
Address terms |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | CLIN INFORMAT PL STAT | 102810 | 0% | 100% | 2 |
| 2 | GLOBAL DEV OPERAT DEV | 102810 | 0% | 100% | 2 |
| 3 | ACOSOG | 68539 | 0% | 67% | 2 |
| 4 | BIOINFORMAT OPERAT SYST | 68539 | 0% | 67% | 2 |
| 5 | CLIN DEV INNOVAT | 68539 | 0% | 67% | 2 |
| 6 | ALDO CELE DACO | 51405 | 0% | 100% | 1 |
| 7 | ANCILLARY PSYCHOSOCIAL SERV | 51405 | 0% | 100% | 1 |
| 8 | ANESTHESIA PAIN MANGEMENT | 51405 | 0% | 100% | 1 |
| 9 | ASIA COHORT CONSORTIUM COORDINATING | 51405 | 0% | 100% | 1 |
| 10 | ASIA PACIFIC J AN REG | 51405 | 0% | 100% | 1 |
Journals |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | CONTROLLED CLINICAL TRIALS | 267016 | 12% | 7% | 71 |
| 2 | CLINICAL TRIALS | 128016 | 7% | 6% | 43 |
| 3 | DRUG INFORMATION JOURNAL | 126610 | 9% | 5% | 52 |
| 4 | THERAPEUTIC INNOVATION & REGULATORY SCIENCE | 51086 | 3% | 5% | 19 |
| 5 | CONTEMPORARY CLINICAL TRIALS | 16140 | 4% | 1% | 21 |
| 6 | CRC CRITICAL REVIEWS IN MEDICAL INFORMATICS | 3670 | 0% | 7% | 1 |
| 7 | ACCOUNTABILITY IN RESEARCH-POLICIES AND QUALITY ASSURANCE | 2522 | 1% | 2% | 3 |
| 8 | METHODS OF INFORMATION IN MEDICINE | 2437 | 2% | 0% | 10 |
| 9 | TRIALS | 2050 | 2% | 0% | 11 |
| 10 | NURSING RESEARCH | 1906 | 2% | 0% | 9 |
Author Key Words |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
LCSH search | Wikipedia search |
|---|---|---|---|---|---|---|---|
| 1 | RISK BASED MONITORING | 781895 | 3% | 89% | 17 | Search RISK+BASED+MONITORING | Search RISK+BASED+MONITORING |
| 2 | DOUBLE DATA ENTRY | 365546 | 1% | 89% | 8 | Search DOUBLE+DATA+ENTRY | Search DOUBLE+DATA+ENTRY |
| 3 | PROTOCOL COMPLEXITY | 308431 | 1% | 100% | 6 | Search PROTOCOL+COMPLEXITY | Search PROTOCOL+COMPLEXITY |
| 4 | ELECTRONIC DATA CAPTURE | 261342 | 3% | 27% | 19 | Search ELECTRONIC+DATA+CAPTURE | Search ELECTRONIC+DATA+CAPTURE |
| 5 | SOURCE DATA VERIFICATION | 231320 | 1% | 75% | 6 | Search SOURCE+DATA+VERIFICATION | Search SOURCE+DATA+VERIFICATION |
| 6 | REMOTE DATA ENTRY | 228981 | 1% | 64% | 7 | Search REMOTE+DATA+ENTRY | Search REMOTE+DATA+ENTRY |
| 7 | LARGE SIMPLE TRIAL | 168229 | 1% | 55% | 6 | Search LARGE+SIMPLE+TRIAL | Search LARGE+SIMPLE+TRIAL |
| 8 | CLINICAL DATA MANAGEMENT SYSTEM | 154215 | 1% | 100% | 3 | Search CLINICAL+DATA+MANAGEMENT+SYSTEM | Search CLINICAL+DATA+MANAGEMENT+SYSTEM |
| 9 | TELEFORM | 154215 | 1% | 100% | 3 | Search TELEFORM | Search TELEFORM |
| 10 | GOOD CLINICAL PRACTICE | 124342 | 3% | 16% | 15 | Search GOOD+CLINICAL+PRACTICE | Search GOOD+CLINICAL+PRACTICE |
Core articles |
The table includes core articles in the class. The following variables is taken into account for the relevance score of an article in a cluster c: (1) Number of references referring to publications in the class. (2) Share of total number of active references referring to publications in the class. (3) Age of the article. New articles get higher score than old articles. (4) Citation rate, normalized to year. |
| Rank | Reference | # ref. in cl. |
Shr. of ref. in cl. |
Citations |
|---|---|---|---|---|
| 1 | MACEFIELD, RC , BESWICK, AD , BLAZEBY, JM , LANE, JA , (2013) A SYSTEMATIC REVIEW OF ON-SITE MONITORING METHODS FOR HEALTH-CARE RANDOMISED CONTROLLED TRIALS.CLINICAL TRIALS. VOL. 10. ISSUE 1. P. 104-124 | 37 | 62% | 6 |
| 2 | OLSEN, R , BIHLET, AR , KALAKOU, F , ANDERSEN, JR , (2016) THE IMPACT OF CLINICAL TRIAL MONITORING APPROACHES ON DATA INTEGRITY AND COST-A REVIEW OF CURRENT LITERATURE.EUROPEAN JOURNAL OF CLINICAL PHARMACOLOGY. VOL. 72. ISSUE 4. P. 399 -412 | 19 | 90% | 1 |
| 3 | VON NIEDERHAUSERN, B , ORLETH, A , SCHADELIN, S , RAWI, N , VELKOPOLSZKY, M , BECHERER, C , BENKERT, P , SATALKAR, P , BRIEL, M , PAULI-MAGNUS, C , (2017) GENERATING EVIDENCE ON A RISK-BASED MONITORING APPROACH IN THE ACADEMIC SETTING - LESSONS LEARNED.BMC MEDICAL RESEARCH METHODOLOGY. VOL. 17. ISSUE . P. - | 21 | 72% | 0 |
| 4 | HULLSIEK, KH , KAGAN, JM , ENGEN, N , GRARUP, J , HUDSON, F , DENNING, ET , CAREY, C , COURTNEY-RODGERS, D , FINLEY, EB , JANSSON, PO , ET AL (2015) INVESTIGATING THE EFFICACY OF CLINICAL TRIAL MONITORING STRATEGIES: DESIGN AND IMPLEMENTATION OF THE CLUSTER RANDOMIZED START MONITORING SUBSTUDY.THERAPEUTIC INNOVATION & REGULATORY SCIENCE. VOL. 49. ISSUE 2. P. 225 -233 | 17 | 77% | 1 |
| 5 | LE JEANNIC, A , QUELEN, C , ALBERTI, C , DURAND-ZALESKI, I , (2014) COMPARISON OF TWO DATA COLLECTION PROCESSES IN CLINICAL STUDIES: ELECTRONIC AND PAPER CASE REPORT FORMS.BMC MEDICAL RESEARCH METHODOLOGY. VOL. 14. ISSUE . P. - | 18 | 72% | 3 |
| 6 | PAVLOVIC, I , KERN, T , MIKLAVCIC, D , (2009) COMPARISON OF PAPER-BASED AND ELECTRONIC DATA COLLECTION PROCESS IN CLINICAL TRIALS: COSTS SIMULATION STUDY.CONTEMPORARY CLINICAL TRIALS. VOL. 30. ISSUE 4. P. 300-316 | 15 | 88% | 38 |
| 7 | JIANG, KY , CAO, X , (2011) DESIGN AND IMPLEMENTATION OF AN AUDIT TRAIL IN COMPLIANCE WITH US REGULATIONS.CLINICAL TRIALS. VOL. 8. ISSUE 5. P. 624 -633 | 15 | 88% | 1 |
| 8 | BAKOBAKI, J , JOFFE, N , BURDETT, S , TIERNEY, J , MEREDITH, S , STENNING, S , (2012) A SYSTEMATIC SEARCH FOR REPORTS OF SITE MONITORING TECHNIQUE COMPARISONS IN CLINICAL TRIALS.CLINICAL TRIALS. VOL. 9. ISSUE 6. P. 777-780 | 13 | 87% | 4 |
| 9 | POGUE, JM , DEVEREAUX, PJ , THORLUND, K , YUSUF, S , (2013) CENTRAL STATISTICAL MONITORING: DETECTING FRAUD IN CLINICAL TRIALS.CLINICAL TRIALS. VOL. 10. ISSUE 2. P. 225 -235 | 15 | 68% | 11 |
| 10 | TIMMERMANS, C , DOFFAGNE, E , VENET, D , DESMET, L , LEGRAND, C , BURZYKOWSKI, T , BUYSE, M , (2016) STATISTICAL MONITORING OF DATA QUALITY AND CONSISTENCY IN THE STOMACH CANCER ADJUVANT MULTI-INSTITUTIONAL TRIAL GROUP TRIAL.GASTRIC CANCER. VOL. 19. ISSUE 1. P. 24 -30 | 11 | 92% | 0 |
Classes with closest relation at Level 1 |