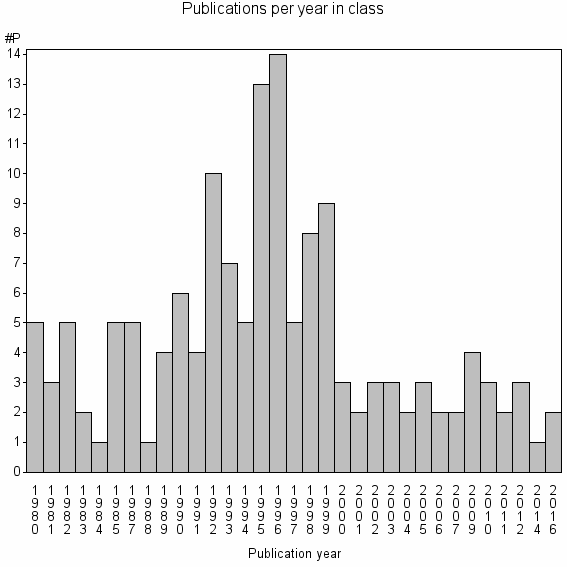
Class information for: |
Basic class information |
| Class id | #P | Avg. number of references |
Database coverage of references |
|---|---|---|---|
| 31862 | 147 | 23.8 | 64% |
Hierarchy of classes |
The table includes all classes above and classes immediately below the current class. |
| Cluster id | Level | Cluster label | #P |
|---|---|---|---|
| 0 | 4 | BIOCHEMISTRY & MOLECULAR BIOLOGY//CELL BIOLOGY//ONCOLOGY | 4064930 |
| 373 | 3 | ENDOGENOUS RETROVIRUS//VIROLOGY//JOURNAL OF VIROLOGY | 32972 |
| 636 | 2 | ENDOGENOUS RETROVIRUS//RETROVIRUS//RETROVIRAL VECTOR | 13733 |
| 31862 | 1 | RT ASSAY//POLY DNP RNA//REVERSE TRANSCRIPTASE INHIBITING ANTIBODY | 147 |
Terms with highest relevance score |
| rank | Term | termType | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|---|
| 1 | RT ASSAY | authKW | 865518 | 3% | 83% | 5 |
| 2 | POLY DNP RNA | authKW | 415449 | 1% | 100% | 2 |
| 3 | REVERSE TRANSCRIPTASE INHIBITING ANTIBODY | authKW | 415449 | 1% | 100% | 2 |
| 4 | REVERSE TRANSCRIPTASE ASSAY | authKW | 324563 | 3% | 31% | 5 |
| 5 | PRODUCT ENHANCED REVERSE TRANSCRIPTASE ASSAY | authKW | 311584 | 2% | 50% | 3 |
| 6 | HOST MEDIATED INVIVO INVITRO ASSAY | authKW | 276965 | 1% | 67% | 2 |
| 7 | 2 FLUOROPOLYNUCLEOLIDES | authKW | 207725 | 1% | 100% | 1 |
| 8 | 5 BROMO 2 DEOXYURIDINE TRIPHOSPHATE | authKW | 207725 | 1% | 100% | 1 |
| 9 | BIOTIN 11 DEOXYURIDINETRIPHOSPHATE | authKW | 207725 | 1% | 100% | 1 |
| 10 | CELLULAR DNA POLYMERASE | authKW | 207725 | 1% | 100% | 1 |
Web of Science journal categories |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | Virology | 2160 | 29% | 0% | 43 |
| 2 | Biochemical Research Methods | 768 | 24% | 0% | 36 |
| 3 | Biotechnology & Applied Microbiology | 579 | 27% | 0% | 40 |
| 4 | Biochemistry & Molecular Biology | 89 | 24% | 0% | 35 |
| 5 | Oncology | 59 | 12% | 0% | 18 |
| 6 | Immunology | 32 | 9% | 0% | 13 |
| 7 | Infectious Diseases | 27 | 5% | 0% | 7 |
| 8 | Microbiology | 24 | 7% | 0% | 10 |
| 9 | Biophysics | 15 | 5% | 0% | 7 |
| 10 | Pharmacology & Pharmacy | 13 | 8% | 0% | 12 |
Address terms |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | H LEE MOFFITT CANC CLIN INVEST PRO | 207725 | 1% | 100% | 1 |
| 2 | H LEE MOFFITT CANC HEMATOL MALIGNA | 207725 | 1% | 100% | 1 |
| 3 | INTERNAL MED INTERDISCIPLINARY ONCOL PR | 207725 | 1% | 100% | 1 |
| 4 | MED BIOCHEM NO 1 JEN AI RD SEC 1 | 207725 | 1% | 100% | 1 |
| 5 | UNITE CONTROLE MEDICAMENTS IMMUNOL | 207725 | 1% | 100% | 1 |
| 6 | ANIM CELL BANK | 103861 | 1% | 50% | 1 |
| 7 | HBIGS | 103861 | 1% | 50% | 1 |
| 8 | MOL BIOL ZBC | 103861 | 1% | 50% | 1 |
| 9 | STABEN | 103861 | 1% | 50% | 1 |
| 10 | UMR 2142 CNRS BIOMERIEUX | 103861 | 1% | 50% | 1 |
Journals |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | JOURNAL OF VIROLOGICAL METHODS | 11919 | 14% | 0% | 20 |
| 2 | OLIGONUCLEOTIDES | 6944 | 2% | 1% | 3 |
| 3 | EXPERIMENTELLE PATHOLOGIE | 3519 | 1% | 2% | 1 |
| 4 | BIOTECHNOLOGY AND APPLIED BIOCHEMISTRY | 2470 | 3% | 0% | 5 |
| 5 | ANTISENSE & NUCLEIC ACID DRUG DEVELOPMENT | 2230 | 1% | 1% | 2 |
| 6 | TRANSFUSION SCIENCE | 1493 | 1% | 0% | 2 |
| 7 | BIOLOGICALS | 1370 | 2% | 0% | 3 |
| 8 | SURVEY OF IMMUNOLOGIC RESEARCH | 1213 | 1% | 1% | 1 |
| 9 | CLINICAL AND DIAGNOSTIC VIROLOGY | 987 | 1% | 0% | 1 |
| 10 | BIOTECHNIQUES | 850 | 3% | 0% | 5 |
Author Key Words |
Core articles |
The table includes core articles in the class. The following variables is taken into account for the relevance score of an article in a cluster c: (1) Number of references referring to publications in the class. (2) Share of total number of active references referring to publications in the class. (3) Age of the article. New articles get higher score than old articles. (4) Citation rate, normalized to year. |
| Rank | Reference | # ref. in cl. |
Shr. of ref. in cl. |
Citations |
|---|---|---|---|---|
| 1 | ODAWARA, F , ABE, H , KOHNO, T , NAGAI-FUJII, Y , ARAI, K , IMAMURA, S , MISAKI, H , AZUMA, H , IKEBUCHI, K , IKEDA, H , ET AL (2002) A HIGHLY SENSITIVE CHEMILUMINESCENT REVERSE TRANSCRIPTASE ASSAY FOR HUMAN IMMUNODEFICIENCY VIRUS.JOURNAL OF VIROLOGICAL METHODS. VOL. 106. ISSUE 1. P. 115-124 | 18 | 82% | 10 |
| 2 | EKSTRAND, DHL , AWAD, RJK , KALLANDER, CFR , GRONOWITZ, JS , (1996) A SENSITIVE ASSAY FOR THE QUANTIFICATION OF REVERSE TRANSCRIPTASE ACTIVITY BASED ON THE USE OF CARRIER-BOUND TEMPLATE AND NON-RADIOACTIVE-PRODUCT DETECTION, WITH SPECIAL REFERENCE TO HUMAN-IMMUNODEFICIENCY-VIRUS ISOLATION.BIOTECHNOLOGY AND APPLIED BIOCHEMISTRY. VOL. 23. ISSUE . P. 95 -105 | 15 | 75% | 28 |
| 3 | FAN, XY , LU, GZ , WU, LN , CHEN, JH , XU, WQ , ZHAO, CN , GUO, SQ , (2006) A MODIFIED SINGLE-TUBE ONE-STEP PRODUCT-ENHANCED REVERSE TRANSCRIPTASE (MSTOS-PERT) ASSAY WITH HEPARIN AS DNA POLYMERASE INHIBITOR FOR SPECIFIC DETECTION OF RTASE ACTIVITY.JOURNAL OF CLINICAL VIROLOGY. VOL. 37. ISSUE 4. P. 305-312 | 13 | 62% | 4 |
| 4 | ZHANG, C , WU, Y , SUN, YN , HONG, C , XIANG, KH , GUO, Y , BARTLAM, M , LOU, ZY , (2010) A NOVEL NON-RADIOACTIVE ASSAY FOR HIV-RT (RDDP) BASED ON PYROSEQUENCING FOR HIGH-THROUGHPUT DRUG SCREENING.PROTEIN & CELL. VOL. 1. ISSUE 3. P. 284-290 | 12 | 57% | 1 |
| 5 | KOTHAPALLI, R , DANYLUCK, GM , BAILEY, RD , LOUGHRAN, TP , (2003) PROBLEMS ASSOCIATED WITH PRODUCT ENHANCEMENT REVERSE TRANSCRIPTASE ASSAY USING BACTERIOPHAGE MS2 RNA AS A TEMPLATE.JOURNAL OF VIROLOGICAL METHODS. VOL. 109. ISSUE 2. P. 203-207 | 11 | 73% | 0 |
| 6 | ANDRE, M , MORGEAUX, S , FUCHS, F , (2000) QUANTITATIVE DETECTION OF RT ACTIVITY BY PERT ASSAY: FEASIBILITY AND LIMITS TO A STANDARDIZED SCREENING ASSAY FOR HUMAN VACCINES.BIOLOGICALS. VOL. 28. ISSUE 2. P. 67 -80 | 14 | 52% | 11 |
| 7 | VOISSET, C , TONJES, RR , BREYTON, P , MANDRAND, B , PARANHOS-BACCALA, G , (2001) SPECIFIC DETECTION OF RT ACTIVITY IN CULTURE SUPERNANTANTS OF RETROVIRUS-PRODUCING CELLS, USING SYNTHETIC DNA AS COMPETITOR IN POLYMERASE ENHANCED REVERSE TRANSCRIPTASE ASSAY.JOURNAL OF VIROLOGICAL METHODS. VOL. 94. ISSUE 1-2. P. 187-193 | 11 | 61% | 9 |
| 8 | SEARS, JF , KHAN, AS , (2003) SINGLE-TUBE FLUORESCENT PRODUCT-ENHANCED REVERSE TRANSCRIPTASE ASSAY WITH AMPLIWAX (TM) (STF-PERT) FOR RETROVIRUS QUANTITATION.JOURNAL OF VIROLOGICAL METHODS. VOL. 108. ISSUE 1. P. 139-142 | 8 | 73% | 13 |
| 9 | PIZZATO, M , ERLWEIN, O , BONSALL, D , KAYE, S , MUIR, D , MCCLURE, MO , (2009) A ONE-STEP SYBR GREEN I-BASED PRODUCT-ENHANCED REVERSE TRANSCRIPTASE ASSAY FOR THE QUANTITATION OF RETROVIRUSES IN CELL CULTURE SUPERNATANTS.JOURNAL OF VIROLOGICAL METHODS. VOL. 156. ISSUE 1-2. P. 1-7 | 12 | 39% | 29 |
| 10 | VERMEIRE, J , NAESSENS, E , VANDERSTRAETEN, H , LANDI, A , IANNUCCI, V , VAN NUFFEL, A , TAGHON, T , PIZZATO, M , VERHASSELT, B , (2012) QUANTIFICATION OF REVERSE TRANSCRIPTASE ACTIVITY BY REAL-TIME PCR AS A FAST AND ACCURATE METHOD FOR TITRATION OF HIV, LENTI- AND RETROVIRAL VECTORS.PLOS ONE. VOL. 7. ISSUE 12. P. - | 15 | 29% | 16 |
Classes with closest relation at Level 1 |