Human Secretome Project
The overall aim of the Secretome project is to produce, purify and characterize the majority of all human secreted proteins to build up a resource of reagents for drug discovery and development.
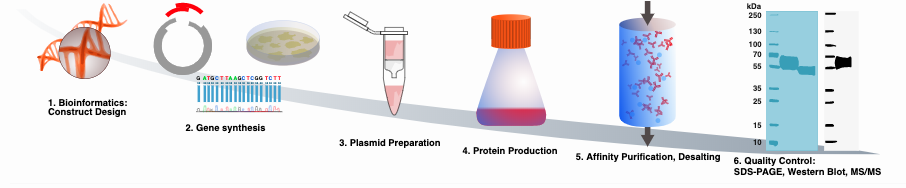
All constructs produced within the Human Secretome Project are synthesized with an N-terminal signal peptide and a C-terminal purification handle. After gene synthesis the constructs are subcloned into an expression vector and sequenced verified prior protein production. In the Human Secretome Project proteins are produced in Chinese Hamster Ovary (CHO) cells using transient protein production. The expression system used is developed by Icosagen Cell Factory. The CHO cells are transfected by electroporation, feeded twice and harvested after 13 days. As a result of the common signal peptide the produced proteins will be secreted into the culture media.
After harvest the proteins are affinity purified using the C-terminal purification handle and a high-throughput protein purification protocol developed in house. The purity and glycosylation patterns of the purified proteins are determined using SDS-PAGE and Western blot. MS/MS is finally used to verify the identity of the protein.
In this high-throughput production platform for full length proteins we have a capacity of up to 50 cultivations, 36 purifications and 30 protein identifications each week.
The proteins can be used for many different applications such as phenotypic assays, immunoassay development, antibody generation and interaction studies.